 Here we have mentioned all the chemical compounds with their chemical formulas. Using ethanol as an example, its condensed structural formula is CH3CH2OH. It omits some bond lines and shows atoms grouped together. The condensed formula is a variation of the structural formula that simplifies it further. It provides a detailed visual representation of a compound’s structure. The structural formula uses lines to represent bonds between atoms and shows how atoms and shows how atoms are arranged in the molecule. (b) Solid calcium hydroxide reacts with aqueous. 
(a) Upon heating, solid magnesium carbonate decomposes into solid magnesium oxide and carbon dioxide gas. Be sure to use the proper symbols to indicate the state of each substance, as given. For example, the molecular formula for hydrogen peroxide is also H2O2. Change these word equations into formula equations and balance them. The molecular formula gives the exact composition of a compound. The molecular formula shows the actual number of atoms of each element in a molecule. For example, the empirical formula for hydrogen peroxide is H2O2. magnesium propanoate would be written (C 2 H 5 COO. It is usual to write the formula of the salts with the -anoate bit first. The empirical formula doesn’t provide the exact number ratio of atoms but shows the relative proportions. acid + metal carbonate salt + water + carbon dioxide. 
The empirical formula informs about the simplest whole-number or the most reduced ratio of elements in a given compound. Simplified structural formula with omitted bond lines. Uses lines to represent bonds and atom arrangement. The molecular formula is the actual number of atoms of each element in a molecule. The empirical formula is the simplest whole-number ratio of elements in a compound. Here we have mentioned some of the common types of chemical formulas used in chemistry to describe and represent different types of compounds and molecules. Under normal combustion/oxidation circumstances, oxygen is the reactant. What's unusual, however, is that magnesium is reactive enough to be combusted and oxidized in a reaction with carbon dioxide: 2Mg +CO2 2MgO + C (1) (1) 2 Mg + CO 2 2 MgO + C. Types of Chemical FormulasĬhemical formulas represent the composition of chemical compounds. Explanations (including important chemical equations) This reaction is a combustion and oxidation. The chemical formulas are essential for describing and communicating the composition of substances in chemistry. For example, H2O is the chemical formula for water, which indicates that it consists of two hydrogen (H) atoms and one oxygen (O) atom bonded together. Subscript numbers indicate the number of atoms of each element in a molecule of the compound. The chemical formula of a compound provides essential information about its composition and structure.Įlemental symbols are abbreviations for the chemical elements present in the compound. It shows the types and numbers of atoms that make up the compounds. The numbers of ions in a formula must give an equal number of positive and negative charges.The chemical formula of a compound is a representation of a chemical composition that provides information about the types and numbers of atoms present in the compound using elemental symbols and subscripts. The formula of an ionic compound can be predicted using the formulae of its ions. Names and formulae of some common ions: Name of ion The number and sign of its electrical charges are shown in superscript text. 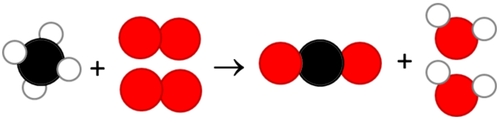
Chemical formulae of ionsĪn ion is a charged particle formed when an atom, or a group of atoms, loses or gains electrons. So for carbon dioxide there are two oxygen atoms for every carbon atom. This shows the number of atoms of this element for every one atom of the first element in the name. In compounds made up of non-metal elements only, the second word of the compound's name starts with mon-, di-, or tri-, eg carbon dioxide. It is not easy to split up a compound into its elements - the only way to do this is in chemical reactions. 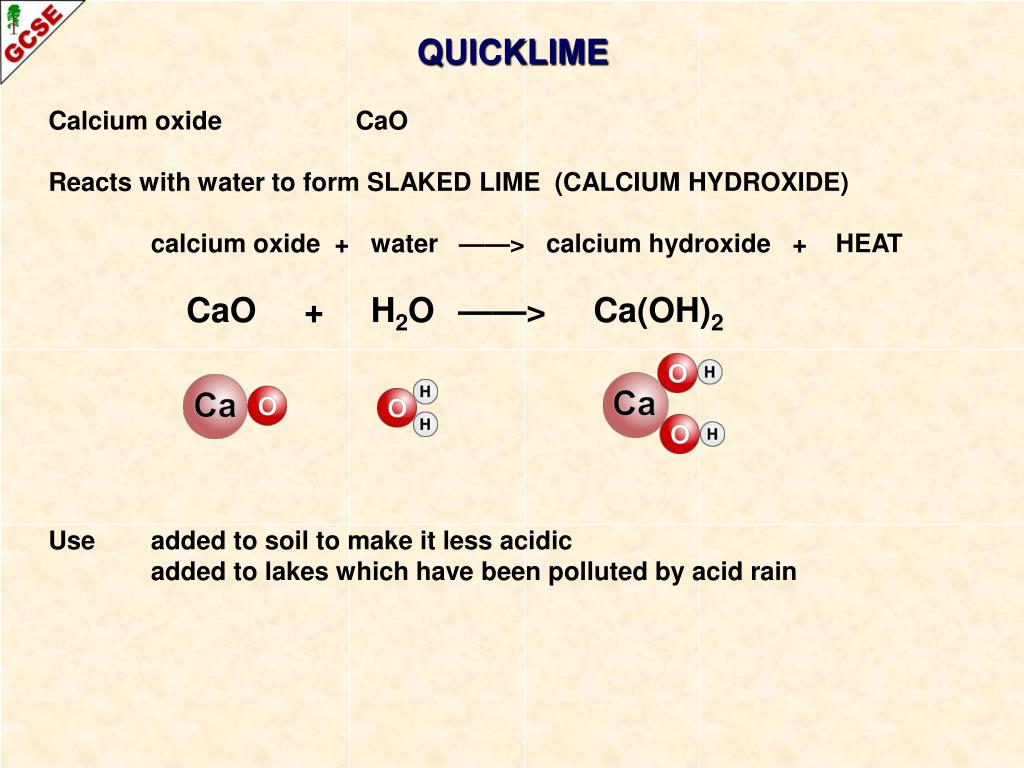
Most chemical reactions involve energy changes. In a chemical reaction, one or more new substances are formed. They can also be formed from their elements in chemical reactions. The subscript number in a formula shows if there is more than one atom of an element. Here are some more examples of compounds and their formulae. This shows that it has one atom of magnesium for every one atom of oxygen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
